Enroflox 100®
(enrofloxacin) Injectable Solution
A sterile, ready-to-use injectable antimicrobial solution that contains enrofloxacin, a broad-spectrum fluoroquinolone antimicrobial agent.
Target Species: Beef cattle, Non-Lactating dairy cattle and Swine
Product Attributes
-
Same active ingredient and dosing regimen as Baytril® 100 (enrofloxacin) Injectable Solution in beef, non-lactating dairy cattle and swine
-
Approved for single dose treatment and control & multi-day treatment options in cattle
-
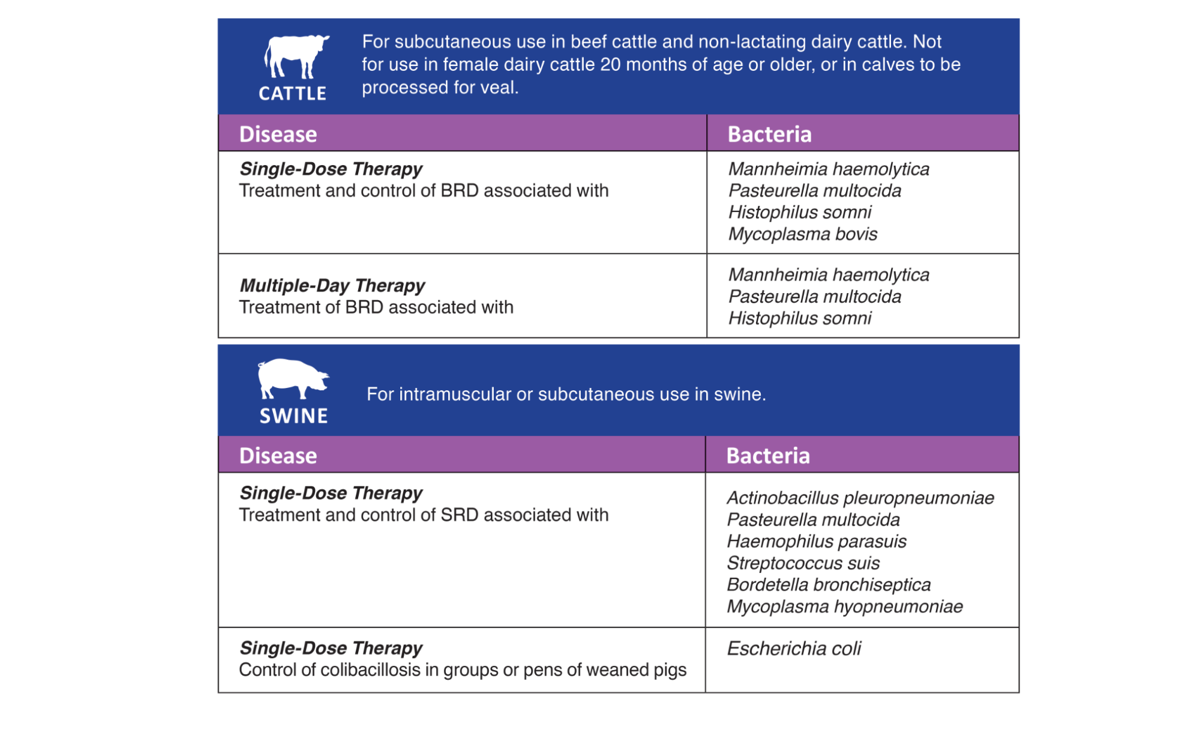
Cattle – Single-Dose Therapy: Enroflox® 100 is indicated for the treatment of Bovine Respiratory Disease (BRD) associated with Mannheimia haemolytica, Pasteurella multocida, Histophilus somni and Mycoplasma bovis in beef and non-lactating dairy cattle; and for the control of BRD in beef and non-lactating dairy cattle at high risk of developing BRD associated with M. haemolytica, P. multocida, H. somni and M. bovis.
-
Cattle – Multi-Day Therapy: Enroflox® 100 is indicated for the treatment of bovine respiratory disease (BRD) associated with Mannheimia haemolytica, Pasteurella multocida and Histophilus somni in beef and non-lactating dairy cattle.
-
Swine: Enroflox® 100 is indicated for the treatment and control of swine respiratory disease (SRD) associated with Actinobacillus pleuropneumoniae, Pasteurella multocida, Haemophilus parasuis, Streptococcus suis, Bordetella bronchiseptica and Mycoplasma hyopneumoniae. Enroflox® 100 is indicated for the control of colibacillosis in groups or pens of weaned pigs where colibacillosis associated with Escherichia coli has been diagnosed.
-
One-dose treatment in pigs
-
Effective, ready to use and easy to inject
-
FDA approved
-
Available in 100 mL, 250 mL and 500 mL vials to fit any size operation
CAUTION:
Federal (U.S.A.) law restricts this drug to use by or on the order of a licensed veterinarian. Federal (U.S.A.) law prohibits the extra-label use of this drug in food-producing animals. To assure responsible antimicrobial drug use, enrofloxacin should only be used as a second-line drug for colibacilosis in swine following consideration of other therapeutic options.
Cattle intended for human consumption must not be slaughtered within 28 days from the last treatment. This product is not approved for female dairy cattle 20 months of age or older, including dry dairy cows. Use in these cattle may cause drug residues in milk and/or calves born to these cows. A withdrawal period has not been established in pre-ruminating calves. Do not use in calves to be processed for veal. To assure responsible antimicrobial drug use, enrofloxacin should only be used as a second-line drug for colibacillosis in swine following consideration of other therapeutic options. Swine intended for human consumption must not be slaughtered within 5 days of receiving a single-injection dose. Use with caution in animals with known or suspected CNS disorders. Observe label directions and withdrawal times. See product labeling for full product information.
-
Active Ingredient(s)
enrofloxacin 100 mg/mL
-
Dosage Form
Ready-to-use injectable solution
-
Indications
Cattle - Single-Dose Therapy: Enroflox® 100 is indicated for the treatment of bovine respiratory disease (BRD) associated with Mannheimia haemolytica, Pasteurella multocida, Histophilus somni and Mycoplasma bovis in beef and non-lactating dairy cattle; and for the control of BRD in beef and non-lactating dairy cattle at high risk of developing BRD associated with M. haemolytica, P. multocida, H. somni and M. bovis.
Cattle - Multiple-Day Therapy: Enroflox® 100 is indicated for the treatment of bovine respiratory disease (BRD) associated with Mannheimia haemolytica, Pasteurella multocida and Histophilus somni in beef and non-lactating dairy cattle.
Swine: Enroflox® 100 is indicated for the treatment and control of swine respiratory disease (SRD) associated with Actinobacillus pleuropneumoniae, Pasteurella multocida, Haemophilus parasuis, Streptococcus suis, Bordetella bronchiseptica and Mycoplasma hyopneumoniae. Enroflox® 100 is indicated for the control of colibacillosis in groups or pens of weaned pigs where colibacillosis associated with Escherichia coli has been diagnosed.
-
Dosage and Administration
DOSAGE
Enroflox® 100 provides flexible dosages and durations of therapy.
Enroflox® 100 may be administered as a single dose for one day for treatment and control of BRD (cattle), for treatment and control of SRD or for control of colibacillosis (swine), or for multiple days for BRD treatment (cattle). Selection of the appropriate dose and duration of therapy for BRD treatment in cattle should be based on an assessment of the severity of the disease, pathogen susceptibility and clinical response.ADMINISTRATION
Cattle:
Single-Dose Therapy (BRD Treatment): Administer, by subcutaneous injection, a single dose of 7.5-12.5 mg/kg of body weight (3.4-5.7 mL/100 lb).Multiple-Day Therapy (BRD Treatment): Administer daily, a subcutaneous dose of 2.5-5 mg/kg of body weight (1.1-2.3 mL/100 lb). Treatment should be repeated at 24-hour intervals for three days. Additional treatments may be given on Days 4 and 5 to animals that have shown clinical improvement but not total recovery.
Single-Dose Therapy (BRD Control): Administer, by subcutaneous injection, a single dose of 7.5 mg/kg of body weight (3.4 mL/100 lb). Examples of conditions that may contribute to calves being at high risk for developing BRD include, but are not limited to, the following:
• Transportation with animals from two or more farm origins.
• An extended transport time with few to no rest stops.
• An environmental temperature change of ≥30°F during transportation.
• A ≥30°F range in temperature fluctuation within a 24-hour period.
• Exposure to wet or cold weather conditions.
• Excessive shrink (more than would be expected with a normal load of cattle).
• Stressful arrival processing procedures (e.g., castration or dehorning).
• Exposure within the prior 72 hours to animals showing clinical signs of BRD. Administered dose volume should not exceed 20 mL per injection site.Table 1 - Enroflox® 100 Dose and Treatment Schedule for Cattle*
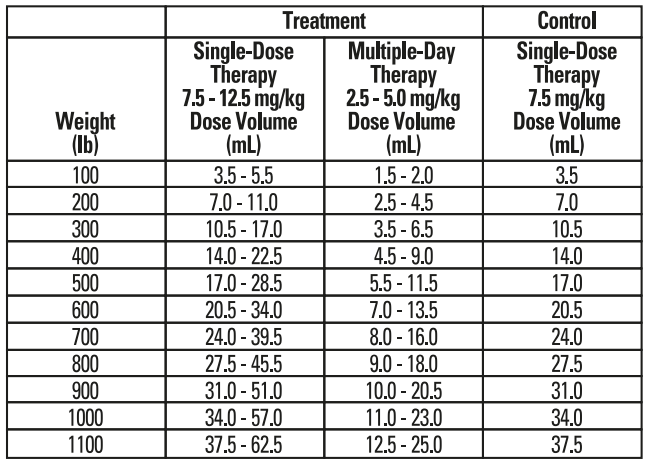
*Dose volumes have been rounded to the nearest 05. mL within the dose range.
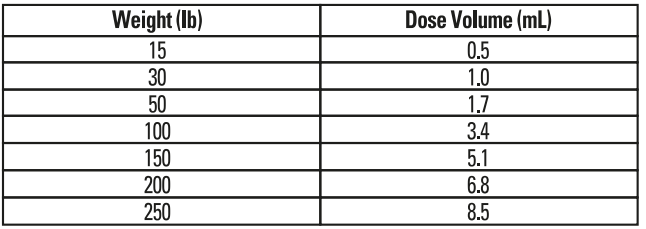
Swine:
Administer, either by intramuscular or subcutaneous (behind the ear) injection, a single dose of 7.5 mg/kg of body weight (3.4 mL/100 lb). Administered dose volume should not exceed 5 mL per injection site.For the control of colibacillosis, administration should be initiated within the first 60 days post-weaning when clinical signs are present in at least 2% of the animals in the group. If no improvement is noted within 48 hours, the diagnosis should be re-evaluated.
Table 2 - Enroflox® 100 Dose Schedule for Swine
Dilution of Enroflox® 100: Enroflox® 100 may be diluted with sterile water prior to injection. The diluted product should be used within 24 hours. Store diluted solution in amber glass bottles between 4-40°C (36-104°F).
Table 3 - Dilution Schedule*
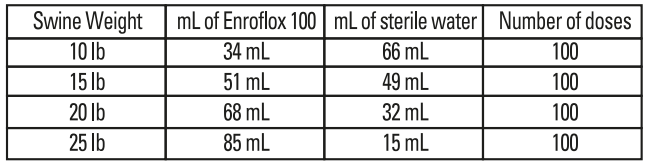
*For 1 mL dose volume from diluted solution.
100 mL vial: Use within 30 days of first puncture and puncture a maximum of 36 times. When using a needed or draw-off spike larger than 16 gauge, discard any remaining product immediately after use.
250 mL and 500 mL vials: Use within 30 days of first puncture. Puncture a maximum of 36 times with a needle or dosage delivery devise 16 gauge or smaller, or 4 times with a draw-off spike 5 mm or smaller. When using a needle larger than 16 gauge, or a draw-off spike larger than 5 mm, discard any remaining product immediately after use.
-
Storage
Protect from direct sunlight. Do not refrigerate or freeze. Store below 77°F (25°C). Precipitation may occur due to cold temperature. To redissolve, warm and then shake the vial.
A Comprehensive Antibiotic Portfolio
Norbrook® now covers five classes of antibiotics.
© 2023 Norbrook Laboratories Limited. The Norbrook logo and Enroflox are registered trademarks of Norbrook Laboratories Limited. Baytril is a registered trademark of Elanco or its affiliates.

Prefer to speak directly to a member of our team?
Norbrook®, Inc.
We are open Mon - Fri 8:00am - 5:00pm CST
Please note: Product information presented on this website is intended only as a brief summary of Norbrook products for your convenience. Not all products or indications are licensed in every country and may be subject to further local variations. For specific product information you should always consult a healthcare professional from your region or visit the local government agency website for the most up to date information. Please see our terms and conditions for further information.