Midamox®
(imidacloprid + moxidectin) for Dogs and Cats
THE AFFORDABLE PARASITE-FIGHTING SOLUTION
Midamox® (imidacloprid + moxidectin) topical solution is bioequivalent to Advantage Multi® and is made with the only combination of active ingredients approved by the FDA to kill circulating heartworm microfilariae in dogs. Midamox® is an effective, affordable choice for protecting pets against heartworm disease, treating flea infestations and treating and controlling several other parasites.
Target Species: Dogs and Cats
The Midamox® price point allows you to:
-
Help your clients save money on their pet’s wellness costs
-
Reduce your inventory costs and improve clinic profit potential
-
Encourage compliance with lower costs
-
Potentially increase the number of pets in your practice that are protected year-round
CAUTION: Federal (U.S.A.) law restricts this drug to use by or on the order of a licensed veterinarian.
Dogs: WARNING: DO NOT ADMINISTER THIS PRODUCT ORALLY. For the first 30 minutes after application ensure that dogs cannot lick the product from application sites on themselves or other treated animals. Children should not come in contact with the application sites for two (2) hours after application. (See Contraindications, Warnings, Human Warnings, and Adverse Reactions for more information.) CONTRAINDICATIONS: Do not use this product on cats.
Cats and Ferrets: WARNINGS: Do not use on sick or debilitated cats or ferrets. Do not use on underweight cats (see ADVERSE REACTIONS). Do not use on cats less than 9 weeks of age or less than 2 lbs. body weight. Do not use on ferrets less than 2 lbs. body weight. PRECAUTIONS: Avoid oral ingestion. HUMAN WARNINGS: Children should not come in contact with the application site for 30 minutes after application.
-
Active Ingredient(s)
Midamox® for Dogs: 10% imidacloprid and 2.5% moxidectin
Midamox® for Cats: 10% imidacloprid and 1% moxidectin
-
Dosage Form and Description
Topical solution: a colorless to yellow, ready-to-use solution packaged in single-dose applicators for topical application.
-
Indications
Midamox® for Dogs is indicated for the prevention of heartworm disease caused by Dirofilaria immitis and the treatment of Dirofilaria immitis circulating microfilariae in heartworm-positive dogs. Midamox® for Dogs kills adult fleas and is indicated for the treatment of flea infestations (Ctenocephalides felis). Midamox® for Dogs is also indicated for the treatment and control of sarcoptic mange caused by Sarcoptes scabiei var. canis. Midamox® for Dogs is also indicated for the treatment and control of the following intestinal parasites.
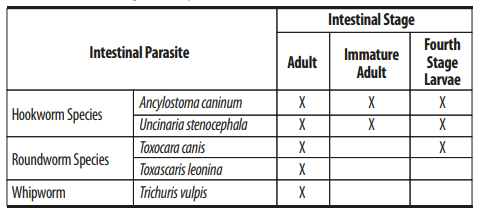
Midamox® for Cats is indicated for the prevention of heartworm disease caused by Dirofilaria immitis. Midamox® for Cats kills adult fleas (Ctenocephalides felis) and is indicated for the treatment of flea infestations. Midamox® for Cats is also indicated for the treatment and control of ear mite (Otodectes cynotis) infestations and the following intestinal parasites:

-
Dosage and Administration
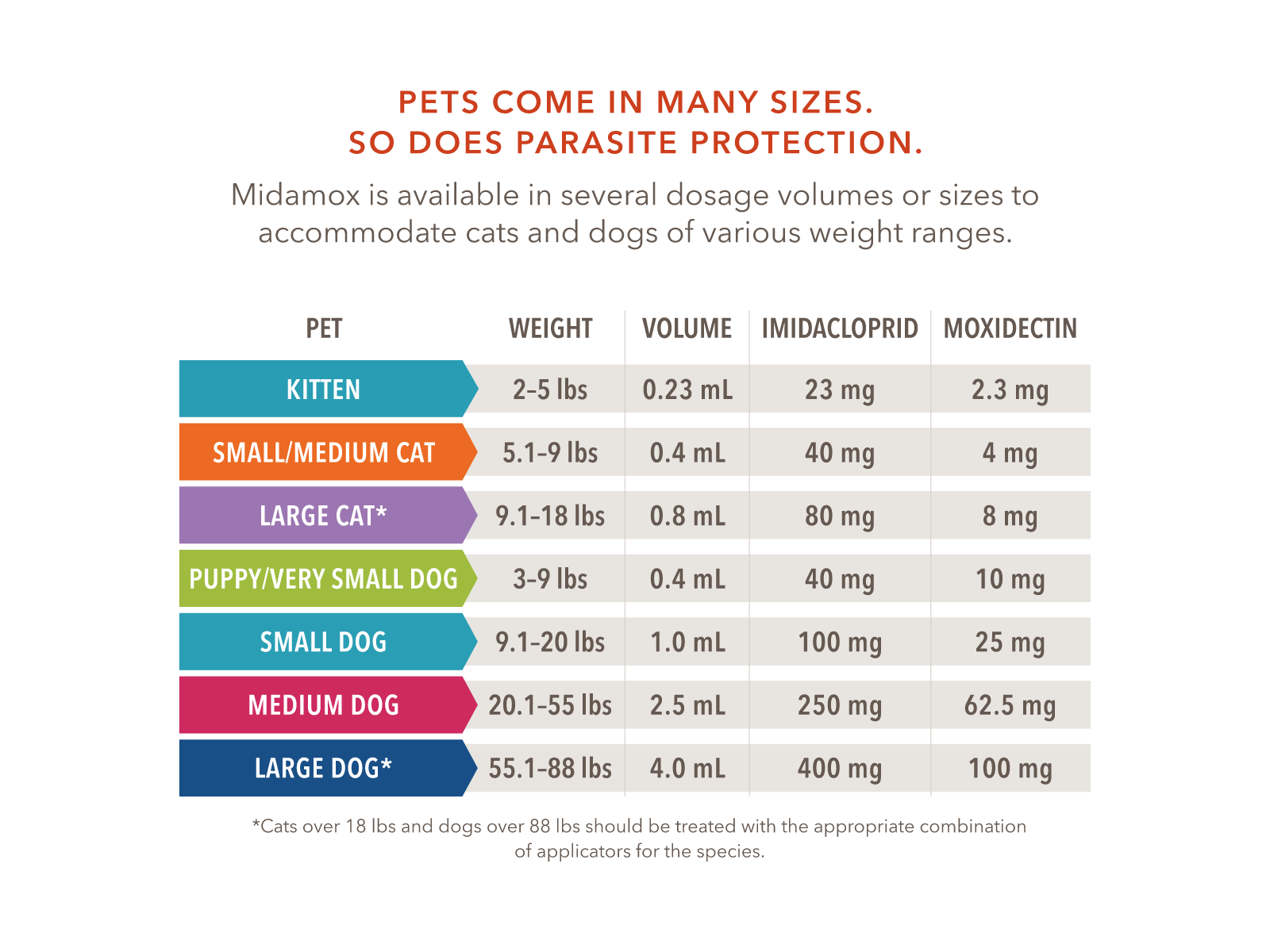
Midamox® offers a range of dosage volumes or sizes to accommodate various weights of cats and dogs.
Once a month topical treatment. Apply to the back of the neck at the base of the head for cats, and between the shoulder blades in dogs. The animal should be standing for easy application. Remove any collar prior to application and replace when product is dry. Part the hair until the skin is visible. Place the tip of the pipette on the skin and squeeze the pipette firmly several times to apply the solution directly on the skin. Avoid pet’s eyes or mouth. See package insert for specific instructions.
-
Storage
Store below 77°F (25°C). Excursions are permitted up to 104°F (40°C) however such exposures should be minimized. Do not remove the applicator from the pouch until ready to use. Do not use after the expiry date which is stated on the carton.
How to apply Midamox® (imidacloprid + moxidectin) on your Dog or Cat
Watch our quick 'how to' video to learn how easy it is to apply Midamox® (imidacloprid + moxidectin) on your dog or cat.
© 2023 Norbrook Laboratories Limited. The Norbrook logo and Midamox are registered trademarks of Norbrook Laboratories Limited. Advantage Multi is a registered trademark of Elanco or its affiliates.
If you are a pet owner looking to purchase Midamox® Topical Solution, please consult with your local veterinarian.

Prefer to speak directly to a member of our team?
Norbrook®, Inc.
We are open Mon - Fri 8:00am - 5:00pm CST
Please note: Product information presented on this website is intended only as a brief summary of Norbrook products for your convenience. Not all products or indications are licensed in every country and may be subject to further local variations. For specific product information you should always consult a healthcare professional from your region or visit the local government agency website for the most up to date information. Please see our terms and conditions for further information.











