Durvet
Enroflox®
(enrofloxacin) Chewable Tablets
The affordable alternative to Baytril® (enrofloxacin) Taste Tabs®. Enroflox® (enrofloxacin) Chewable Tablets are a fluoroquinolone antibiotic that manages diseases associated with bacteria susceptible to enrofloxacin.
Target Species: Dogs and Cats
Enroflox® contains the same active ingredient as the pioneer product, making it an effective treatment against susceptible dermal, respiratory and urinary tract infections in dogs and dermal infections in cats. These pork liver-flavored tablets are double-scored for flexible, accurate dosing and offer significant savings over the pioneer product.
Product Attributes
-
Indications for both dogs and cats
-
Same active ingredient and dosing regimen as pioneer product
-
Flexible daily dosing for dogs of 5-20 mg/kg (2.27 – 9.07 mg/lb) of body weight
-
Daily dose for cats at 5 mg/kg (2.27 mg/lb) of body weight
-
Concentration dependent and bactericidal
-
Kills a broad range of susceptible Gram-positive and Gram-negative bacteria
-
Less expensive than Baytril® Taste Tabs®, offering significant savings and improved clinic profit potential
-
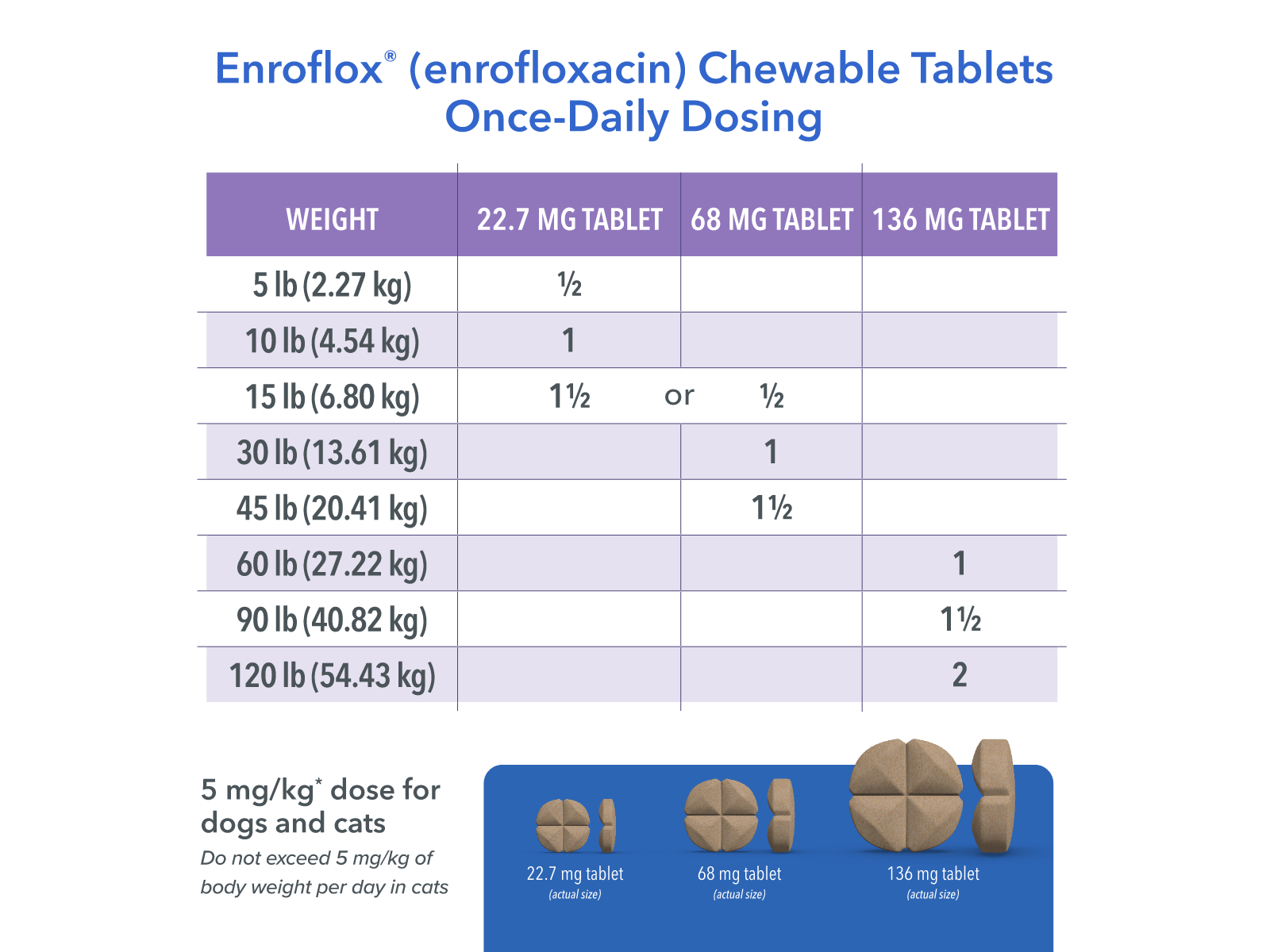
Available in 22.7 mg, 68 mg and 136 mg strengths
-
Uniform 50 count and 200 count bottles across all 3 strengths
CAUTION: Federal (U.S.A.) law restricts this drug to use by or on the order of a licensed veterinarian. In rare instances, use of this product in cats has been associated with retinal toxicity. Do not exceed 2.27 mg/lb of body weight per day in cats. Observe label directions and see product labeling for full product information.
-
Active Ingredient(s)
enrofloxacin
-
Dosage Form and Description
Chewable tablet: a brown ‘clover’ design tablet in three strengths (22.7 mg, 68 mg and 136 mg), double scored. The tablets can be divided into quarters or halves if required.
-
Indications
Enroflox® Chewable Tablets (enrofloxacin) are indicated for the management of diseases associated with bacteria susceptible to enrofloxacin. Enroflox® Chewable Tablets are indicated for use in dogs and cats.
-
Dosage and Administration
Dogs: Administer orally at a rate to provide 5-20 mg/kg (2.27 to 9.07 mg/lb) of body weight. Selection of a dose within the range should be based on clinical experience, the severity of disease, and susceptibility of the pathogen. Animals which receive doses in the upper-end of the dose range should be carefully monitored for clinical signs that may include inappetence, depression, and vomition.
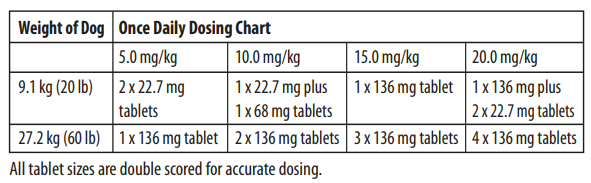
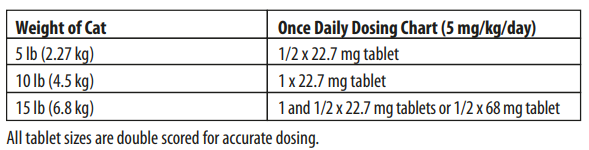
Cats: Administer orally at 5 mg/kg (2.27 mg/lb) of body weight. The dose for dogs and cats may be administered either as a single daily dose or divided into two (2) equal daily doses administered at twelve (12) hour intervals. The dose should be continued for at least 2-3 days beyond cessation of clinical signs, to a maximum of 30 days.
-
Storage
Dispense tablets in tight containers only. Enroflox® Chewable Tablets (enrofloxacin) should be stored at or below 77°F (25°C). Use half and quarter tablets within 90 days.
© 2023 Norbrook Laboratories Limited. The Norbrook logo and Enroflox are registered trademarks of Norbrook Laboratories Ltd. Baytril and Taste Tabs are registered trademarks of Elanco or its affiliates.
Order Enroflox® Chewables Today
Select your preferred distributor.
First Veterinary Supply
HSB Veterinary Supply, Inc.
Midwest Veterinary Supply
Penn Veterinary Supply
If you are a pet owner looking to purchase Enroflox® Chewable Tablets, please consult with your local veterinarian.

Prefer to speak directly to a member of our team?
Norbrook®, Inc.
We are open Mon - Fri 8:00am - 5:00pm CST
Please note: Product information presented on this website is intended only as a brief summary of Norbrook products for your convenience. Not all products or indications are licensed in every country and may be subject to further local variations. For specific product information you should always consult a healthcare professional from your region or visit the local government agency website for the most up to date information. Please see our terms and conditions for further information.










